 How many moles are present in 1.00 × 10 23 PF 3 molecules?.How many moles are present in 2.09 × 10 22 Ti atoms?.How many moles are present in 3.55 × 10 24 Pb atoms?.How many molecules are present in 0.336 mol of acetylene (C 2H 2)?.How many molecules are present in 2.509 mol of H 2S?.How many atoms are present in 0.0665 mol of K?.How many atoms are present in 4.55 mol of Fe?. :max_bytes(150000):strip_icc()/PeriodicTablecolor-58b5d9105f9b586046df7d8a.jpg)
These relationships give us plenty of opportunities to construct conversion factors for simple calculations. In each of these moles of substances, there are 6.022 × 10 23 units: 6.022 × 10 23 atoms of H, 6.022 × 10 23 molecules of H 2 and H 2O, 6.022 × 10 23 units of NaCl ions. A single unit of NaCl has a mass of 58.45 u NaCl has a molar mass of 58.45 g.A molecule of H 2O has a mass of about 18.01 u 1 mol H 2O has a mass of 18.01 g.Elemental hydrogen exists as a diatomic molecule, H 2.The mass of a hydrogen (H) atom is 1.0079 u the mass of 1 mol of H atoms is 1.0079 g.(The term molar is the adjective form of mole and has nothing to do with teeth.) Sometimes these masses are called molar masses to emphasize the fact that they are the mass for 1 mol of things. The atomic mass of an element is the number of grams in 1 mol of atoms of that element, while the molecular mass of a compound is the number of grams in 1 mol of molecules of that compound. Previously we defined atomic and molecular masses as the number of atomic mass units per atom or molecule. How many grams are there, say, in 1.50 × 10 25 atoms of carbon? This is a one-step conversion:ġ.50 \times 10^īut it also goes beyond carbon. We can use this equality as a conversion factor between the number of atoms of carbon and the number of grams of carbon. This means that exactly 12 g of C has 6.022 × 10 23 atoms: The mole unit allows us to express amounts of atoms and molecules in visible amounts that we can understand.įor example, we already know that, by definition, a mole of carbon has a mass of exactly 12 g. A mole of a substance has the same mass in grams as one unit (atom or molecules) has in atomic mass units. Why is the mole unit so important? It represents the link between the microscopic and the macroscopic, especially in terms of mass. One mole of carbon atoms would make a cube that is 1.74 cm on a side, small enough to carry in your pocket. A mole is a lot of things-but atoms and molecules are very tiny. A mole of pennies stacked on top of each other would have about the same diameter as our galaxy, the Milky Way. A mole of sand would fill a cube about 32 km on a side. If everyone on earth (about 6 billion people) counted one bill per second, it would take about 3.2 million years to count all the bills. Suppose you had a mole of dollar bills that need to be counted. How many molecules are present in 4.61 × 10 −2 mol of O 2? Answer To determine the total number of atoms, we have: 
Also, because we know that there are three atoms in each molecule of H 2O, we can also determine the number of atoms in the sample. The definition of a mole is an equality that can be used to construct a conversion factor. How many molecules are present in 2.76 mol of H 2O? How many atoms is this? Solution The numerical value of things in a mole is often called Avogadro’s number ( N A), which is also known as the Avogadro constant, after Amadeo Avogadro, an Italian chemist who first proposed its importance. Although the number of things in a mole is known to eight decimal places, it is usually fine to use only two or three decimal places in calculations. These things can be atoms, or molecules, or eggs however, in chemistry, we usually use the mole to refer to the amounts of atoms or molecules. But a mole is a much larger number of things. Understand that a mole means a number of things, just like a dozen means a certain number of things-twelve, in the case of a dozen. Experimental measurements have determined that this number is very large: A mole (mol) is a number of things equal to the number of atoms in exactly 12 g of carbon-12. We need a unit of amount that relates quantities of substances on a scale that we can interact with.Ĭhemistry uses a unit called mole. What we need is a way to deal with macroscopic, rather than microscopic, amounts of matter. 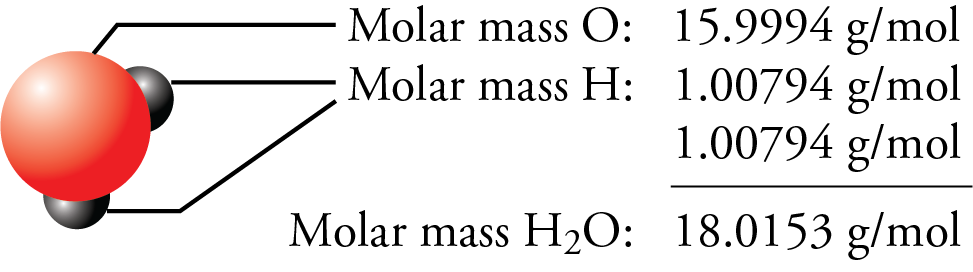
Yet we don’t typically deal with substances an atom or a molecule at a time we work with millions, billions, and trillions of atoms and molecules at a time. So far, we have been talking about chemical substances in terms of individual atoms and molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
